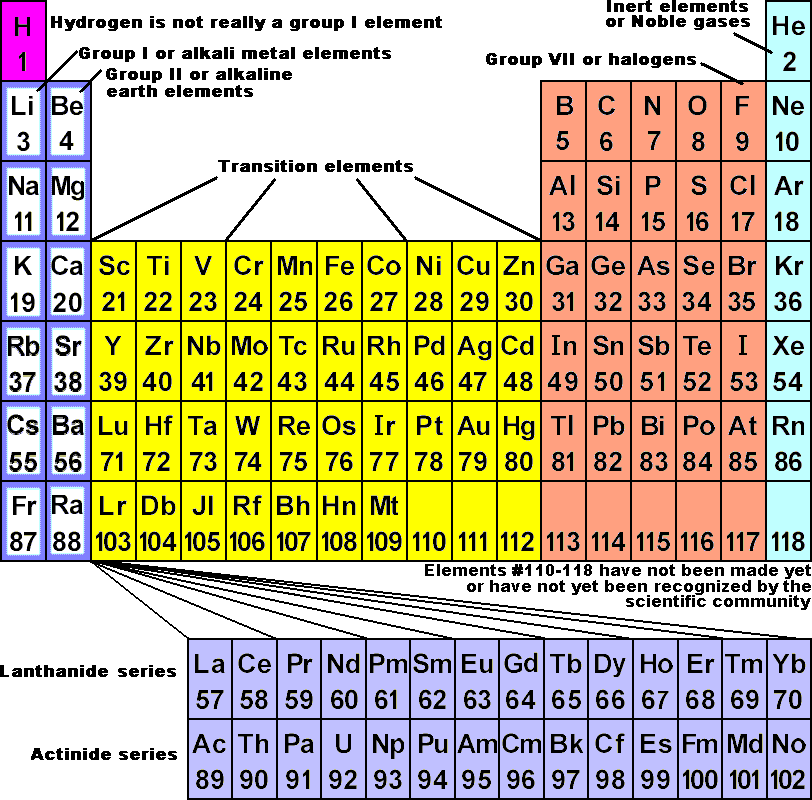
Elements with similar properties are grouped together. Mendeleev originally arranged elements in ascending order of atomic weight, one beneath the other. Mendeleev’s law states that “the Physical and Chemical Properties of the Elements are Periodic Functions of Their Atomic Weights.” What was Mendeleev’s Elements Arranging Method? What exactly is Mendeleev’s Periodic Law? The element displaying the repeat of the properties was placed in the first slot of this new template. As soon as the qualities of the components were discovered to be duplicated, he stopped that row and began a new one below it. Mendeleev devised a row of elements arranged in ascending atomic weight order. He arranged the elements in the periodic table so that those with similar qualities were grouped together in the same vertical columns. After studying the properties of elements, Mendeleev determined that they were related to atomic mass in a periodic pattern. At the time of Mendeleev, only 63 elements were known. Elements were organised in Mendeleev’s periodic table based on their fundamental properties, atomic mass, and chemical properties. There have been several periodic tables developed, but the Mendeleev periodic table is the most important.įollowing the rejection of Newlands Octave Law in 1869, the Mendeleev Periodic Table was developed. They also share properties with both the metals and the nonmetals.Mendeleev Periodic Table:- Dmitri Ivanovich Mendeleev, a Russian scientist, was the most essential contributor to the early development of the periodic table. They have low electrical conductivity that increases with temperature. The elements at the boundary between the metallic elements (grey elements) and nonmetal elements (green elements) are metalloid in character (pink elements). These elements are insulators, poor heat conductors, and can exist in different phases at room temperature (brittle solid, liquid, or gas). Moving from left to right across a period, the elements have greater non-metallic character. The further down a given group, the elements have an increased metallic character: they are good conductors of both heat and electricity, solids at room temperature, and shiny in appearance. Within the main group elements ( Groups 1-2, 13-18), there are some general trends that we can observe. The arrangement of the periodic table allows the elements to be grouped according to their chemical properties. The atomic number appears above the symbol for the element and the approximate atomic mass appears below it. The periodic table shows the atomic mass and atomic number of each element. Looking at carbon, for example, its symbol (C) and name appear, as well as its atomic number of six (in the upper left-hand corner) and its atomic mass of 12.11. In addition to providing the atomic number for each element, the periodic table also displays the element's atomic mass. 
If you look at a periodic table, you will see the groups numbered at the top of each column from left to right starting with 1 and ending with 18. In the periodic table the elements are organized and displayed according to their atomic number and are arranged in a series of rows (periods) and columns (groups) based on shared chemical and physical properties. Devised by Russian chemist Dmitri Mendeleev (1834–1907) in 1869, the table groups elements that, although unique, share certain chemical properties with other elements. The different elements are organized and displayed in the periodic table. Carbon, hydrogen, oxygen, and nitrogen make up approximately 96% of the mass in a human body. There are 25 elements believed to play an active role in human health. Pure chemical substances consisting of only one type of atom with a defined set of chemical and physical properties.Įlements of the human body arranged by percent of total mass Properties found in compound structures that are different from those of the individual components and would not be predicted based on the properties of the individual components. The periodic table continues to expand today as heavier and heavier elements are created in laboratories around the world.Ī tabular chart of the chemical elements according to their atomic numbers so that elements with similar properties are in the same column.As one moves down a column or across a row, there are some general trends for the properties of the elements.The periodic table is a listing of the elements according to increasing atomic number that is further organized into columns based on similar physical and chemical properties and electron configuration.Combining elements creates compounds that may have emergent properties.Living creatures consist mainly of carbon, hydrogen, oxygen, and nitrogen (CHON). All matter is made from atoms of one or more elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
